Truvada and other TDF-based HIV drugs can cause bone loss or osteoporosis. These side effects of Truvada and TDF can lead to a sharply increased risk of broken bones or bone fractures. While the warning label for Truvada mentions this risk, it downplays its severity. As a result, many people who have taken TDF-based HIV drugs like Truvada have suffered:
- Osteopenia, or a mildly decreased bone density,
- Osteoporosis, or a bone density that is low enough to be medically problematic,
- Osteomalacia, or bones that have softened due to kidney problems caused by TDF drugs,
- Bone fractures,
- Broken bones, and
- Kidney and renal failure.
In this article, we will discuss:
- 1. Truvada and TDF
- 2. Bone loss and bone density conditions
- 3. Medical studies link TDF and Truvada to bone loss
- 4. Truvada and osteomalacia
- 5. HIV patients are at particular risk for bone loss and fractures
- 6. Lawsuits against Gilead for bone loss injuries
1. Truvada and TDF
Truvada is the brand name of a pill-based HIV treatment made by Gilead Sciences, Inc. It was approved by the U.S. Food and Drug Administration (FDA) in 2004.1
Truvada is a prophylactic treatment for HIV. By preventing HIV cells from spreading once they are in the body, Truvada can treat, but cannot cure, HIV or AIDS. Truvada is a popular treatment for people who are at risk of contracting HIV, like people who are in intimate relationships with someone who is HIV-positive.
Truvada is a combination of two drugs:
- Emtricitabine, and
- Tenofovir disoproxil fumarate, or TDF.
TDF is a very toxic drug. Worse, because it is poorly absorbed in the body it has to be taken in high doses. The toxicity of TDF can cause a variety of side effects. However, people who take Truvada or other TDF-based HIV drug are often willing to deal with those side effects to avoid contracting HIV.
2. Bone loss and bone density conditions
Bone loss is the medical condition of bones deteriorating and becoming less dense. As bone density drops, they weaken and can break or fracture more easily.
Densities in different bones change, though, so measurements are often conducted on different bones:
- Femoral neck, which is the most common place for a hip fracture,
- Overall density of the complete hip,
- Spine, at the first four vertebrae in the lower back, and
- Total body bone density.
These measurements are often done using dual-energy X-ray absorptiometry, also known as either a DEXA or a DXA test. While these special X-ray exams are not perfect, they do produce results that are accurate to approximately 1.5%.2
The results of these DXA tests can be given objectively, in grams of bone mineral per cubic centimeters, as well as subjectively, in terms of standard variation from a normal, healthy bone. These subjective measurements can use either of two different scores:
- T Score, which provides the standard variation from a patient’s bone density compared to a young adult at peak bone density and who is the same gender, or a
- Z Score, which provides the standard variation from the patient’s bone density compared to the average density of someone of the same gender, age, and weight of the patient.
For both scores, a zero means a patient’s bones are exactly as dense as those of either a healthy young adult of the same gender, in the case of T Scores, or the average person of the same age, weight, and gender, for Z Scores. Positive scores in either test mean the patient’s bones are denser. Negative scores mean the patient’s bones are less dense.
Doctors recognize a spectrum for bone density problems, based on a patient’s T Scores:
- Between 0 and -1 is below average, but not bad enough to be a medical condition,
- Between -1 and -2.5 is osteopenia; a low, but not yet medically problematic, bone density, and
- Below -2.5, which is diagnosed as osteoporosis.3
2.1. Osteopenia
Osteopenia is a bone density condition where a patient’s bones are between 1 and 2.5 standard deviations below peak bone density. While it is not considered a severe medical condition, doctors do see it as a precursor to osteoporosis.
There is a debate in the medical community about whether osteopenia is worth treating in aging women who are especially likely to develop the condition.4 However, for the younger, often more active men who are likely to be taking Truvada, preventing osteopenia from developing into osteoporosis becomes more important.
While the risks are relatively low, there is an increased chance that someone with osteopenia will suffer the following injuries or symptoms:
- Bone fractures,
- Broken bones,
- Bone pain, and
- Weakness.
2.2. Osteoporosis
As bone densities drop to the level of osteoporosis, the chances of a broken bone or a fracture increase dramatically because of the weakened bone structure.
There are an estimated 9.9 million people who suffer from osteoporosis.5 However, most people who have osteoporosis do not know they have it until they break or fracture a bone. The most common bones to break because of osteoporosis include:
- Wrists,
- Forearms,
- Hips,
- Ribs, and
- Spinal vertebrae.
While these fractures are rarely fatal, the long-term consequence can be – especially among the elderly. One study found that adults who fractured a hip were between 8 and 36% more likely to die within a year than similar adults who did not break their hip.6
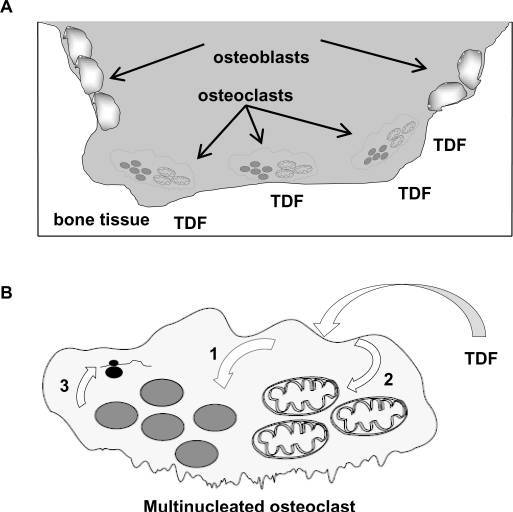
3. Medical studies link TDF and Truvada to bone loss
Numerous medical studies have linked a decrease in bone density to taking Truvada or other TDF-based drugs. Some cases of TDF-related bone loss are severe enough to amount to osteoporosis.7
Even the early clinical trials that Gilead performed when it first released its TDF drugs for HIV treatment suggested that TDF could cause bone loss. These trials were randomized and compared TDF drugs with the existing HIV treatment at the time, stavudine. The results of the trials found that TDF decreased bone density in the spine by 2.2% over three years, while stavudine only led to a 1.0% decrease.8
However, it was not until Gilead Sciences, Inc. began replacing the TDF in its HIV drugs with tenofovir alafenamide fumarate (TAF) that evidence began pouring out about how dangerous TDF could be to a patient’s bone structure. It was at this time that Gilead stopped touting how safe Truvada and its TDF drugs were, and began pushing patients and doctors into using its new TAF drugs.9 In addition to Gilead’s new financial interest in showing that TDF was less safe than its new drugs, the amount of time Truvada and other TDF drugs had been on the market had provided a large sample size to show the toll the drugs took on patients’ bone density.
Most of those studies found that TDF caused between 2.2% and 3.6% bone loss in the spine, and between 2.4% and 4.0% bone loss in the hip.10
Another study drew a distinction between those who had only a low exposure to TDF drugs and those who had high exposures and compared the bone densities of each group over a period of 48 weeks. The group with low exposure to TDF only lost 2.63% of their bone density in the spine, while those with high levels of exposure lost 3.61%. The results were even more damning at the femoral neck, where low exposure patients lost 2.41% of their bone density, but those with high exposure to TDF drugs lost 3.91% over the 48 weeks.11
According to another study, which tracked 56,660 people, TDF exposure resulted in an 8% increased risk of bone fracture, while high exposure to the drug was associated with a 16% increased risk of fracture.12
The connection between TDF drugs like Truvada and bone loss was strengthened by another study, which found that people who take Truvada actually begin to gain bone density back once they stop taking TDF drugs. These patients tended to return to their baseline bone density levels within a year of being off the drug.13
4. Truvada and osteomalacia
In addition to causing bone density problems, TDF drugs can also cause osteomalacia, a medical term for the softening of bones due to changes in the bone’s metabolism.14 Osteomalacia is different from osteopenia or osteoporosis because the bones are softened, rather than lost. Additionally, unlike osteopenia and osteoporosis, which are caused by the toxic effects of TDF directly on a patient’s bone structure, osteomalacia is indirect damage to the bone caused by TDF’s effects on a patient’s kidneys.
One of the major side effects of TDF-based HIV drugs is kidney damage and renal failure. When the kidneys fail, one complication is the accumulation of several different chemicals in the bloodstream – chemicals that the kidney used to be able to distill and expel from the body, but now cannot. One of these chemicals is phosphate. Phosphate is crucial in the process that regenerates old or deteriorated bone structures. When phosphate levels are altered after kidney failure, the bone’s metabolism is thrown out of sync: Not enough bone is created to replace the bone that is decaying. As more bone decays than regenerates, they soften and become more prone to fracturing.
As a result, patients taking TDF-based drugs to treat their HIV are at risk of suffering both bone density problems and softened bones, each of which increases the likelihood of suffering a broken bone.
5. HIV patients are at particular risk for bone loss and fractures
Bone loss caused by TDF-based HIV drugs like Truvada is especially dangerous because people who suffer from HIV already tend to have lower bone densities than other people.
Some of this independent bone loss is due to the nature of the HIV virus, which can compromise the bone structure of the people who have contracted it. The bone loss caused by HIV can increase with the severity of the infection.
Other environmental causes of decreased bone density are also more likely to be present in someone who is HIV-positive, like:
- Illegal drug use,
- Poverty, and
- General poor health.
As a result, one study estimated that up to 90% of people with HIV have bone densities that are low enough to be classified as osteoporosis or as osteopenia.15 Another medical journal article estimated that HIV-positive individuals were 60% more likely than normal to suffer a broken bone or fracture.16
6. Lawsuits against Gilead for bone loss injuries
The injuries caused by the bone loss that was brought about by Truvada have led to hundreds of lawsuits being filed against Gilead. These lawsuits claim that Gilead failed to adequately warn doctors and patients of the risks. They also claim that Gilead withheld the safer TAF drug from the market to maximize the company’s profits.
The TDF lawsuits seek punitive damages against Gilead to hold them accountable for their actions, as well as compensation for the victims’:
- Medical expenses,
- Pain and suffering,
- Lost wages, and
- Loss of consortium.
However, Truvada is not the only brand-name drug that Gilead Sciences, Inc. produces that relies on TDF to treat HIV. In addition to the Truvada lawsuits, legal claims have been filed against Gilead for the bone loss caused by each one of its TDF-based pills, as well as some of its newer TAF drugs, including:
Because these lawsuits involve victims with similar injuries, they are presently being consolidated into an MDL (multidistrict litigation) out of the Northern District of California (federal court). An MDL is where similar lawsuits are consolidated to speed up litigation and a settlement. The Truvada MDL is called In Re. Tenofovir Disoproxil Fumarate Products Liability Litigation, MDL 2881.
References:
- FDA – Approval Letter for Truvada (August 2, 2004).
- See Bilsborough JC, Greenway K, Opar D, Livingstone S, Cordy J, Coutts AJ, “The accuracy and precision of DXA for assessing body composition in team sport athletes,” Journal of Sports Sciences 32(19):1821-8 (June 10, 2014).
- World Health Organization, “Prevention and Management of Osteoporosis,” (2003).
- Nissa Simon, “Are Women Being Overtreated for Bone Loss?” AARP Bulletin (March 27, 2009).
- Wright NC, Looker AC, Saag KG, Curtis JR, Delzell ES, Randall S, Dawson‐Hughes B, “The Recent Prevalence of Osteoporosis and Low Bone Mass in the United States Based on Bone Mineral Density at the Femoral Neck or Lumbar Spine,” Journal of Bone and Mineral Research 29(11):2520-26 (November 2014).
- Abrahamsen B, van Staa T, Ariely R, Olson M, Cooper C, “Excess mortality following hip fracture: a systematic epidemiological review,” Osteoporosis International 20(10):1633-50 (October 2009).
- See generally, Grigsby IF, Pham L, Mansky LM, Gopalakrishnan R, Mansky KC, “Tenofovir-associated bone density loss,” Therapeutics and Clinical Risk Management 6:41-7 (February 2, 2010) and Grant PM, Cotter AG, “Tenofovir and bone health,” Current Opinion in HIV and AIDS 11(3):326-32 (May 2016).
- Gallant JE, et al., “Efficacy and safety of tenofovir DF vs stavudine in combination therapy in antiretroviral-naive patients: a 3-year randomized trial,” Journal of the American Medical Association 292(2):191-201 (July 14, 2004).
- See Melody Petersen, “Patients sue Gilead, saying drug company intentionally delayed safer HIV medicine,” Los Angeles Times (May 9, 2018).
- See note 8.
- Havens PL et al., “Decline in Bone Mass With Tenofovir Disoproxil Fumarate/Emtricitabine Is Associated With Hormonal Changes in the Absence of Renal Impairment When Used by HIV-Uninfected Adolescent Boys and Young Men for HIV Preexposure Prophylaxis,” Clinical Infectious Diseases 64(3):317-25 (February 1, 2017).
- Bedimo R, Maalouf NM, Zhang S, Drechsler H, Tebas P, “Osteoporotic fracture risk associated with cumulative exposure to tenofovir and other antiretroviral agents,” AIDS 26(7):825-31 (April 24, 2012).
- Glidden DV, et al., “Recovery of Bone Mineral Density Following Discontinuation of Tenofovir-Based HIV Pre-Exposure Prophylaxis,” Journal of Acquired Immune Deficiency Syndromes 76(2):177-82 (October 1, 2017).
- Grant PM, Cotter AG, “Tenofovir and bone health,” Current Opinion in HIV and AIDS 11(3):326-32 (May 2016).
- Brown TT, McComsey GA, King MS, Qaqish RB, Bernstein BM, da Silva BA, “Loss of bone mineral density after antiretroviral therapy initiation, independent of antiretroviral regimen,” Journal of Acquired Immune Deficiency Syndromes 51(5):554-61 (August 15, 2009).
- Shiau S, Broun EC, Arpadi SM, Yin MT, “Incident fractures in HIV-infected individuals: a systematic review and meta-analysis,” AIDS 27(12):1949-57 (July 31, 2013).
