Ethicon is a hernia mesh company and is a part of the Johnson & Johnson family of companies. Ethicon first received U.S. Food and Drug Administration (FDA) approval for its hernia mesh in 2010. The mesh, called Physiomesh, has resulted in serious hernia mesh complications in patients and is currently the subject of many federal lawsuits. The mesh is used to treat various types of hernias. Physiomesh is the subject of a recall in many European countries and was the subject of a market withdrawal here in the U.S.
There are nearly 2000 Physiomesh lawsuits filed in the U.S. in Federal courts. In 2017, these cases were consolidated in the Northern District of Georgia and are set for trial in 2018. High rates of complications have been associated with Ethicon products, and your case could be one of them.
The personal injury attorneys at the Shouse Law Group are here to represent injured clients and inform you about the risk and complications associated with Ethicon hernia mesh products.
- 1. What is hernia mesh?
- 2. What is Physiomesh?
- 2.1 What is it made of?
- 3. How was Physiomesh approved by the FDA?
- 4. Was Physiomesh recalled in the U.S.?
- 4.1 What is the difference between a market withdrawal and a recall?
- 5. Are there any side effects or complications?
- 5.1 What is a seroma?
- 5.2 What is mesh migration?
- 5.3 What is chronic pain?
- 5.4 Can I get an infection from Physiomesh?
- 6. Can I be compensated for my injuries?
- 7. What do I do if I was injured by Physiomesh?
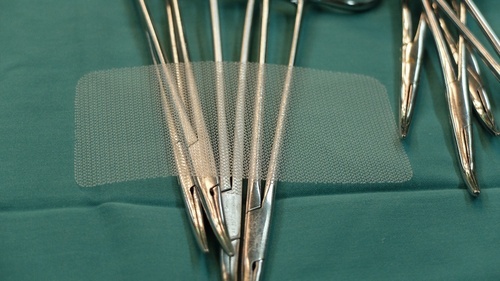
1. What is hernia mesh?
Hernia mesh is a screen-like medical mesh which provides support to damaged and weakened tissue caused by a hernia. A hernia is a bulging of an organ or tissue through an abnormal opening.
Hernia mesh is intended to prevent the re-occurrence of your hernia. Defective hernia mesh leads to thousands of patients suffering complications every year. There are many types and classifications of hernia mesh which a surgeon may use, each with its own benefits and complications.
2. What is Physiomesh?
Physiomesh is a hernia mesh designed primarily for repair of ventral hernias. Like other types of hernia mesh, it is a kind of netting used to stabilize the abdominal area damaged by the hernia.
The netting is coated with a chemical called polypropylene which is absorbed by the body. After the body absorbs these coatings, the Physiomesh plastic remains.
2.1 What is it made of?
Physiomesh is made from a plastic-like material called polypropylene, meaning it is a synthetic mesh. Some studies have shown that synthetic meshes are more likely to result in complications after hernia mesh surgery as compared to hernia mesh created with organic materials. 1
3. How was Physiomesh approved by the FDA?
Physiomesh was approved by the FDA through the 510(K) Premarket Approval Program. This means that this hernia mesh did not go through rigorous medical testing to determine whether it was effective or safe. Ethicon was only required to show that Physiomesh was as safe and effective as similar devices on the market. 2 Ethicon compared Physiomesh to the Proceed hernia patch, a patch it also manufactures.
This program is highly criticized and has allowed defective mesh models to enter the market and be used on patients.
4. Was Physiomesh recalled in the U.S.?
Technically, the answer to this question is no. While Physiomesh was recalled in other countries, in the U.S. it instituted a “market withdrawal” after it had established a replacement product called Physiomesh Open.
Ethicon purposely chose to leave a product it knew to be dangerous on the market so it would not lose money. Only after establishing its new product did it institute the market withdrawal. This exposed many people to the known complications associated with Physiomesh.
4.1 What is the difference between a market withdrawal and a recall?
A recall can be issued by a company’s own choice, an FDA request, or an FDA order to recall a product. It requires safety alerts to be issued to doctors, patients, and hospitals when a recall is issued.
A market withdrawal allows a company to remove the product from the market without issuing a safety alert. This is allowed when the product has a “minor violation” not subject to FDA legal action. Considering the many and often severe side effects Physiomesh has caused in patients, the market withdrawal was not enough to protect the public.
5. Are there any side effects or complications?
There are many different side effects and complications which can result from a doctor’s use of defective Physiomesh or from the doctor’s own negligence.
5.1 What is a seroma?
A seroma is a collection of fluid, called serum, that builds up under the surface of the skin. They can develop where surgical incisions were made in the body or where bodily tissue was removed. These commonly occur after a significant surgical procedure such as a hernia repair and can be the result of defective hernia mesh.
Physiomesh has been shown to cause serious seromas as compared to two other products and was not as effective at integrating itself with the body and surrounding tissues. 3
5.2 What is mesh migration?
Physiomesh has been shown to cause mesh migration, where the mesh moves over time and can cause serious complications. As a result of this movement, studies have shown that as high as 20% of hernia patients treated with Physiomesh suffered a recurrence of their hernia. 4
5.3 What is chronic pain?
Pain after surgery for a short period is normal as your body heals. Severe pain or pain which lasts for longer than 3 months (chronic pain) is not normal and may be caused by defective Physiomesh.
5.4 Can I get an infection from Physiomesh?
Many patients develop infections after surgery with Physiomesh. Many patients develop an allergic reaction to the coating used on the plastic. Others develop infections as a result of organ perforation caused by their hernia mesh.
6. Can I be compensated for my injuries?
If you have suffered injuries as the result of defective Physiomesh, you may be able to file a hernia mesh lawsuit to recover:
- Past and future medical bills
- Lost wages or loss of earning capacity
- Pain and suffering
- Punitive damages (in certain circumstances).
6. What do I do if I was injured by Physiomesh?
If you or someone you know has been harmed by a doctor, nurse, or other healthcare provider’s negligence or by a defective medical product, contact our personal injury lawyers today for a free consultation. Call us or fill out our online contact form on this page.
References
- MDPI. Past, Present and Future of Surgical Meshes: A Review
- FDA. Premarket Notification 510(K)
- NCBI. Tissue integrations and inflammatory reaction.
- NCBI. Comparison of two different concepts of mesh and fixation technique.
